
By Ana Kerridge
In 1781, biophysicist Luigi Galvani accidentally discovered that a frog’s leg would twitch when simultaneously touched by a steel scalpel and brass hook. The mechanism found to be responsible for this led his rival, physicist Alessandro Volta to invent the world’s first electrochemical cell (Veritasium, 2025). This became the basis of most modern-day battery technology - including nickel-cadmium batteries, largely discarded due to their toxicity, lead-acid batteries, and of course, lithium-ion batteries (LIBs) (Whittingham, 2019).
The Electrochemistry
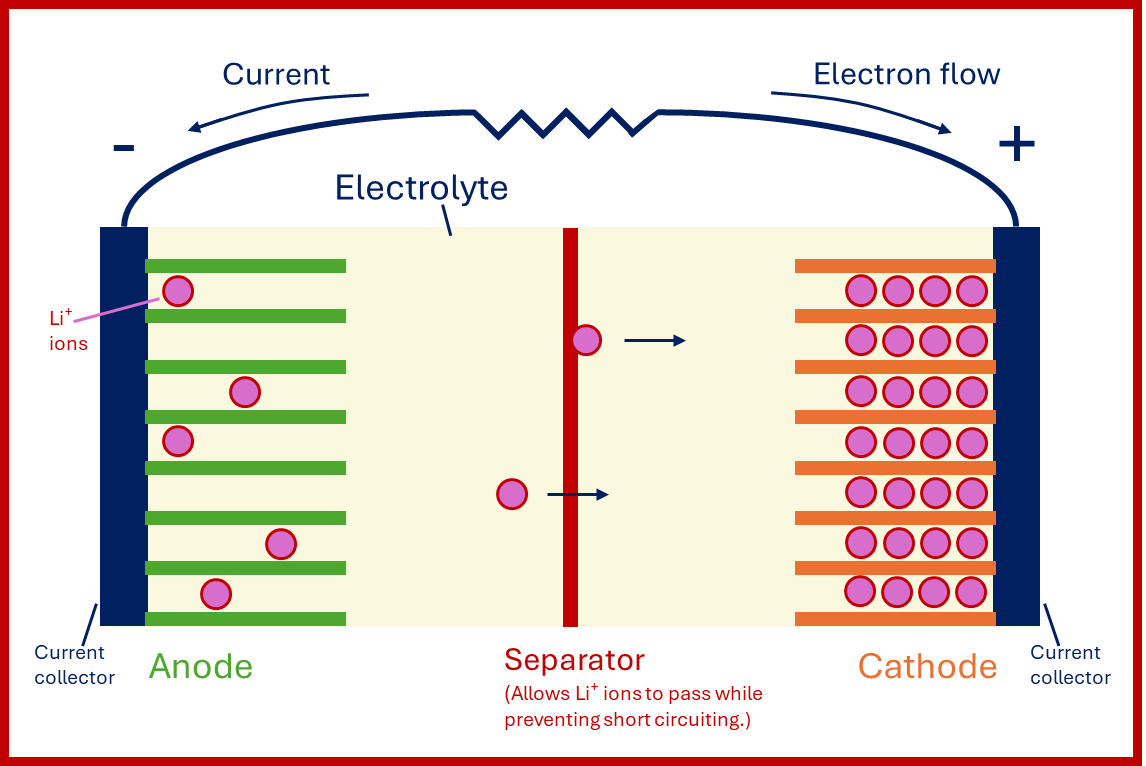
Figure 1: Original diagram of discharging lithium-ion cell. Note that it is convention in electrical diagrams to show flow of current as opposite in direction to that of electrons. Using (Chawla et al., 2019; Krause et al., 2024).
In electrochemical cells (individual units inside batteries), chemical reactions occur at two electrodes: at the anode, electrons (and positive ions, here Li⁺) are released, and at the cathode, accepted. While positive ions move through a liquid electrolyte and separator, the electrons flow through an external circuit; electrical current has been generated!

Figure 2: Example chemical reactions, simplified. Reaction 1 occurs at the anode, here LiC₆ – oxidation occurs (electrons are released). Reaction 2 occurs at the cathode, here LiCoO₂ – reduction occurs (electrons are gained). (GeeksforGeeks, 2023)
While in conventional cells, electrode materials are chemically converted and degraded after repeated cycling, LIBs use “intercalation” - Li⁺ ions slide in/out of a solid electrode’s layered structure. Li⁺ ions are easily reabsorbed during the reverse process (wherein the system is recharged, storing energy until we next need it via discharging), increasing reversibility, cycle life, and efficiency (Battery Burn Book, 2025).
The Process
The word “lithium” originates from the Greek word for stone, λιθος (lithos) (Vanderkrogt, 2026), and although the element can be mined from rock, it is actually more commonly extracted from lithium rich salt brine, pumped up from underground. Water from this reserve evaporates under the sun, leaving a mixture of heavier compounds from which purified lithium compounds are extracted (Anderson, 2023). This method consumes less energy, and releases approximately 70% less carbon dioxide per ton of lithium than mining (MIT Climate Portal Writing Team, 2024). However, it demands huge amounts of water (often in already water-scarce areas), and decreases soil quality, harming local ecosystems and arable land.
In mining lithium, we can continue advancing science, but not without a toll on the environment. Vegetation must often be cleared, vast amounts of carbon dioxide are released near local communities, and toxic chemicals can contaminate local rivers (Anderson, 2023).
Mitigating inevitable impacts relies heavily on regulations, however this is not always clear-cut. Mining company Rio Tinto is working closely with the government in Serbia. Despite their own “extensive tests” showing that ‘boron is the primary element [they] need to manage’ (Rio Tinto, n.d.), the official threshold for the safe concentration of boron in drinking water increased from 0.3mg/L to 1.0mg/L (Federal Republic of Yugoslavia, 1998; Republic of Serbia, 2019). The WHO’s health-based recommendation is 0.5mg/L, reflecting animal studies showing that high intake impaired foetal development, and testicular and kidney function (Baboo, 2021). A 2025 survey estimated that over 60% of Serbs disapprove of the implementation of Rio Tinto’s mines (Vukadinović, 2025). Concerns over threats to safe water, air, cultural sites, and more, have sparked national protests as well as global gatherings - even here in London! (Todorović, 2021).

Figure 3: Original photo of a Serbian protest badge. It reads ‘We will not give away the Jadar!’, referring to the region after which the lithium-containing mineral, jadarite, gets its name (Whitfield et al., 2007)
The Positive and Negative Sides
Batteries, especially ones with high efficiency such as LIBs, are crucial in the green transition. They can store energy from renewable, but intermittently available sources, such as wind and solar power. In a bid to decrease global carbon emissions and optimise energy security, this reduces pressure to exploit highly pollutant fossil fuels - a rapidly depleting resource. Electric vehicles and industrial and home devices can be powered by batteries, without emitting carbon dioxide into the atmosphere (Anderson, 2023).
In addition to issues surrounding mineral extraction explored above, LIBs carry another major downside. Flammable electrolyte, high current, and separator issues, can cause their infamous explosions. We have already seen very rare incidents be fatal, including on planes and at landfill sites. A 2018 investigation by California waste management facilities found that 40% of their fires, which release toxic gases, were caused by LIB failures (United States Environmental Protection Agency, 2021). Although the probability of a single LIB failing is extremely low, the device’s prevalence means that statistically, everyone will witness this at least once in their lifetime! (Veritasium, 2025)
Another major cost behind LIBs is a humanitarian one: Children as young as seven work long days in extremely harsh, dangerous conditions in the Democratic Republic of the Congo, which produces half of the world’s cobalt – a metal needed for common LIB batteries. It is then exported, further refined and processed, before companies including Apple, Samsung, and Volkswagen integrate it into their batteries (Amnesty International, 2016).
Current Climate for LIBs
Two centuries after its invention, the battery is ever more important. Certain social changes could alleviate demand, such as reducing individuals’ reliance on cars, or improving urban planning altogether. Simultaneously, researchers can work on improving lifetime, efficiency, and sustainability of existing and novel batteries.
To provide for the current demand for LIBs, we can recycle their components, reducing the need for harmful, expensive extraction from depleting sources. Recovery rates for key metals can be as high as 93%! (Ma et al., 2025)
Another avenue of research, adopted by the engineering department at UCL, involves replacing the liquid electrolyte with solid. This improves efficiency and longevity of the LIB, as well as reducing its flammability. (Chawla et al., 2019)
Moving away from lithium entirely, zinc-air, zinc-manganese, and sodium-ion batteries are among several alternatives being developed today. Relative to LIBs, all three rely on inexpensive, highly abundant materials, and show promising energy-storage performance. (Lithium Ion News, 2022; Nazir et al., 2024; Zhang et al., 2017)
Galvani and his frog helped launch electrochemistry, now crucial to our transition away from fossil fuels. By improving battery performance, recycling lithium-ion cells, developing alternatives, and tightening regulations at extraction sites, we can support this shift while minimising impacts on local communities and ecosystems.
References:
Veritasium (2025). The Perfect Battery Material Is Dangerous. [online] YouTube. Available at: https://www.youtube.com/watch?v=AGglJehON5g.
Whittingham, M. (2019). The Origins of the Lithium Battery. [Lecture] Available at: https://www.nobelprize.org/uploads/2019/10/whittingham-lecture.pdf.
fig 1. Chawla, N., Bharti, N. and Singh, S. (2019). Recent Advances in Non-Flammable Electrolytes for Safer Lithium-Ion Batteries. [Journal] Research Gate. Available at: https://www.researchgate.net/publication/330798113_Recent_Advances_in_Non-Flammable_Electrolytes_for_Safer_Lithium-Ion_Batteries.
Krause, T., Nusko, D., Pitta Bauermann, L., Vetter, M., Schäfer, M. and Holly, C. (2024). Methods for Quantifying Expansion in Lithium-Ion Battery Cells Resulting from Cycling: A Review. [online] Energies. 17(7), p.1566. doi:https://doi.org/10.3390/en17071566.
GeeksforGeeks. (2023). Electrochemistry Cells and Batteries. [online] GeeksforGeeks. Available at: https://www.geeksforgeeks.org/chemistry/electrochemistry-cells-and-batteries/.
Battery Burn Book (2025) Intercalation & Conversion. [online] Battery Burn Book. Available at: https://www.batteryburnbook.com/learn/intercalation-and-conversion?utm_source.
MIT Climate Portal Writing Team (2024). How is lithium mined? [online] climate.mit.edu. Available at: https://climate.mit.edu/ask-mit/how-lithium-mined.](https://climate.mit.edu/ask-mit/how-lithium-mined).
Vanderkrogt.net. (2026). Lithium - Elementymology & Elements Multidict. [online] Available at: https://elements.vanderkrogt.net/element.php?sym=Li.
Rio Tinto (n.d.). The Jadar Project Mine waste and processing residue. [online] Available at: file:///C:/Users/User/Downloads/rt-jadar-mine-waste-processing-residue.pdf.
Federal Republic of Yugoslavia (1998) Regulation on the hygienic safety of drinking water [Pravilnik o higijenskoj ispravnosti vode za piće]. Official Gazette of the Federal Republic of Yugoslavia [Službeni list SRJ]. 42/98; 44/99.
Republic of Serbia (2019) Regulation on the hygienic safety of drinking water [Правилник о хигијенској исправности воде за пиће]. Official Gazette of the Republic of Serbia [Службени гласник Републике Србије]. 28/19.
Baboo, P. (2021). Boron removal in drinking water. [online] Global Scientific Journal GSJ Publications. Available at: file:///C:/Users/User/Downloads/BORONREMOVALINDRINKINGWATER.pdf.
Vukadinović, Đ. (2025) Istraživanje javnog mnjenja: stavovi građana o rudniku litijuma. Nova srpska politička misao. Available at: http://www.nspm.rs.
Todorović, I. (2021). Masses block roads in Serbia in protest against lithium mining, controversial laws. [online] Balkan Green Energy News. Available at: https://balkangreenenergynews.com/masses-block-roads-in-serbia-in-protest-against-lithium-mining-controversial-laws/.
Whitfield, P.S., Page, Y.L., Grice, J.D., Stanley, C.J. and Gordon (2007). LiNaSiB3O7(OH) – novel structure of the new borosilicate mineral jadarite determined from laboratory powder diffraction data. Acta Crystallographica Section B-structural Science. doi:https://doi.org/10.1107/S0108768107010130/lm5008sup3.pdf.
Anderson, K. (2023). The Harmful Effects of our Lithium Batteries. [online] Greenly.earth. Available at: https://greenly.earth/en-gb/blog/industries/the-harmful-effects-of-our-lithium-batteries.
United States Environmental Protection Agency (2021). An Analysis of Lithium-ion Battery Fires in Waste Management and Recycling. [online] Available at: https://www.epa.gov/system/files/documents/2021-08/lithium-ion-battery-report-update-7.01_508.pdf.
Amnesty International (2016). Exposed: Child labour behind smart phone and electric car batteries. [online] Amnesty International. Available at: https://www.amnesty.org/en/latest/news/2016/01/child-labour-behind-smart-phone-and-electric-car-batteries/.
Ma, X., Meng, Z., Bellonia, M.V., Spangenberger, J., Harper, G., Gratz, E., Olivetti, E., Arsenault, R. and Wang, Y. (2025). The evolution of lithium-ion battery recycling. Nature Reviews Clean Technology, [online] 1(1), pp.75–94. doi:https://doi.org/10.1038/s44359-024-00010-4.
Lithium Ion News. (2022). Fifty years later: the unlikely birth of the lithium-ion battery. [online] Available at: https://lithiumionnews.com/fifty-years-later-the-unlikely-birth-of-the-lithium-ion-battery/
Nazir, G., Rehman, A., Lee, J.-H., Kim, C.-H., Gautam, J., Heo, K., Hussain, S., Ikram, M., AlObaid, A.A., Lee, S.-Y. and Park, S.-J. (2024). A Review of Rechargeable Zinc–Air Batteries: Recent Progress and Future Perspectives. Nano-micro letters, 16(1). doi:https://doi.org/10.1007/s40820-024-01328-1.
Zhang, N., Cheng, F., Liu, J., Wang, L., Long, X., Liu, X., Li, F. and Chen, J. (2017). Rechargeable aqueous zinc-manganese dioxide batteries with high energy and power densities. Nature Communications, 8(1). doi:https://doi.org/10.1038/s41467-017-00467-x.
Share this article
Written by
Scientists say 40-year dispute over Earth’s inner core is finally settled
By Ariella Morris A 40-year scientific dispute over the Earth’s inner core may finally be over, as researchers reveal new evidence explaining how iron atoms arrange themselves at the centre of the planet. Writing in the Journal of Geophysical Research: Solid Earth this year, the team reports that iron
The Unreliable Fabric of Reality
Einstein’s relativity reshaped our view of the universe, showing that space and time shift with motion and gravity. This piece reveals how time slows, lengths change, and simultaneity fades, uncovering the physics behind everyday tech like GPS and why observers never share the same moment.
Are we alone in space?
Scientists suspect Mars was once habitable, but radiation and oxidants erase biological traces. New tests exposing DNA on stones to simulated Martian radiation show that full sequences degrade, yet fragments can survive long enough for taxonomic identification, aiding the search for past life.
