The Origin of Disease from the Womb: Developmental Programming
The extent to which a foetus adapts to the outside environment before birth is phenomenal; but how perfect are these adaptations in preventing disease later in life? By Madeleine Hjelt.
The extent to which a foetus adapts to the outside environment before birth is phenomenal; but how perfect are these adaptations in preventing disease later in life? By Madeleine Hjelt.

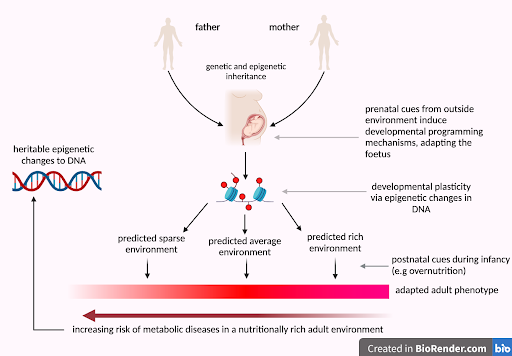
What if I told you that your height, the age you started puberty, and your risk of cardiovascular disease were all factors determined by your mother’s environment during pregnancy? Her nutrition, stress levels, prenatal supplement intake - to name some of the factors - all influence the intrauterine environment and provide outside cues to the fetus [1]. Certain genes are then upregulated or downregulated via epigenetic markers (little chemical tags which bind to DNA molecules directly), adapting the baby to enter either a calm or a high-stress environment. During this stage, physiological factors such as organ growth, metabolic planning and cardiovascular health are determined [2].
Human evolution depends on these adaptations; if a baby is born into a stressful environment, lacking biological preparations would limit survival and prevent the passing down of beneficial genes to future generations [3]. However, in modern day society where threats such as starvation and natural predators are rare, a baby’s biological preparation can mismatch their environment, which has been proven to cause disease. ‘Developmental Programming’ is the term to describe biological adaptations to predicted outside environments. These adaptations allow for human evolution, but are they necessary in today’s society?
During certain critical periods of fetal development, the baby’s ‘plastic’ brain adapts to maternal cues. If the mother is stressed and hungry, wouldn’t it be assumed that the baby will be as well once born? Poor maternal nutrition has been proven to cause low birth weights via Intrauterine Growth Restriction (IUGR), which increases the risk of type II diabetes later in life [4]. Evidence shows that fetal nutrition and oxygenation through the placenta is a large determinant for low birth weights [5]. To compensate for undernutrition, the selective redirection of energy sources to vital organs such as the heart and brain must occur to ensure optimal fetal development. This, however, is at the expense of other tissues such as the insulin secreting β-cells of the pancreas. Such conditions lead to uncontrolled blood sugar levels and predisposes individuals to type II diabetes. Post-birth, affected individuals may exhibit enhanced energy conservation in low-nutrient environments, as observed in the 1943 Siege of Stalingrad, where the ‘small and thin man’ was more likely to survive starvation most likely due to adaptations to lower calorie intake and slower metabolism [4].
However, if babies adapted to famine are born into an environment with high food availability, the risk of obesity and cardiovascular disease skyrockets. Referred to as the ‘thrifty phenotype’ [4], such phenotypes were observed in the Dutch Hunger Winter in World War II: babies born to mothers facing famine, living off 400-800 calories per day, were developmentally programmed to adapt to malnutrition, resulting in smaller placentas and relatively low birth weights. However, when food availability increased post-famine, a mismatch occurred between their adapted physiology and the new environment, causing detrimental outcomes, including a higher risk of obesity, diabetes and coronary artery disease later in life [6]. Factors such as the timing of famine exposure during gestation and gender of the fetus also influenced malnutrition effects later in life. Exposure during the in utero window was found to cause greater effects than exposure during childhood, and due to faster prenatal development in females, males were found to experience more frequent and debilitating health conditions as a result [7].
This is called the Evolutionary Mismatch Hypothesis: when environmental stressors mismatch those to which humans are evolutionarily adapted to, disease occurs [8]. For instance, in cases of maternal malnutrition, the body’s adaptations to low food availability during fetal development may not align with the abundance of food in the true environment, resulting in health complications, as seen in the Dutch Hunger Winter study. These mismatches can persist through multiple generations. For example, British children of South Asian ancestry often have a higher risk of type II diabetes than those of European ancestry, due to historically lower birth weights, a result of adaptive responses to past food scarcity. This leads to hereditary epigenetic modifications which induces higher insulin resistance, causing smaller statures and higher visceral adiposity amongst South Asian children. [9]
In today’s rapidly advancing society, with information overload from social media frying our dopamine receptors, and blue light from our screens altering our circadian rhythms, how will our biochemistry keep up? Studies are already emerging on the technological revolution and climate change’s impact on developmental programming, stating that making phone calls for more than 30 minutes a day is linked to negative effects on fetal development [11] and that extreme heat during pregnancy can cause preterm birth and stillborn fetuses [12]. Predicting the long term effects on our physiology and genetics therefore becomes complex. Mismatches between the predicted and true environment at birth are inevitable, meaning that disease may be inevitable too. Whether our biology can adequately adapt to these global changes remains uncertain, highlighting the need for ongoing research and attention on this subject.
Article written by Madeleine Hjelt
Cover photo credit: Unsplash



